San Francisco, April 29, 2026 – Laguna Biotherapeutics, Inc., a clinical-stage biotechnology company specializing in novel live bacterial therapeutics, announced today that the U.S. Food and Drug Administration (FDA) has cleared its Investigational New Drug (IND) application for LGNA-100. The IND clearance grants a “safe to proceed” status for the company’s lead clinical candidate from its QUAIL platform.
First-in-Class Immunotherapy for High-Risk Leukemia
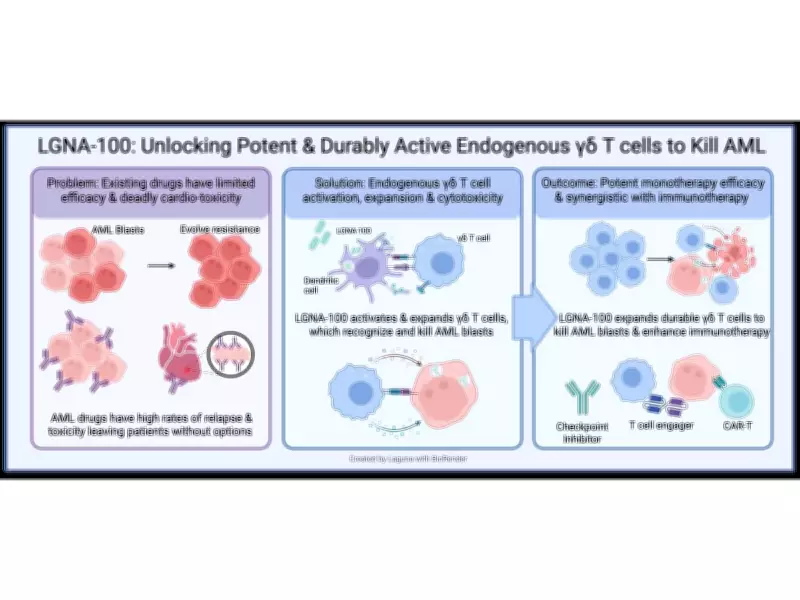
LGNA-100 is a first-in-class, attenuated live bacterial immunotherapy designed to harness the immune system’s evolved response to Listeria. It aims to robustly and durably expand and activate endogenous γδ T cells, which can directly kill cancer cells while also enhancing existing immunotherapies. The Phase 1 first-in-human study will evaluate LGNA-100 in patients with high-risk leukemia following hematopoietic stem cell transplantation (HSCT) to prevent leukemic relapse.
Clinical Rationale and Platform Validation
“The IND clearance of our first clinical study is a defining moment and transformative milestone for Laguna as we transition into a clinical-stage company,” said Jonathan Kotula, Ph.D., CEO of Laguna. “Our goal is to create systems-level therapies for complex diseases. With LGNA-100 and the QUAIL platform, we are taking a fundamentally new approach to selectively stimulate innate T cells to improve long-term outcomes for pediatric patients with high-risk leukemia.”
The clinical rationale for the QUAIL platform builds on decades of research into the human γδ T cell response to Listeria and the protective role of γδ T cells against leukemia recurrence following HSCT. A presentation detailing this rationale will be given at the ISCT 2026 Annual Meeting on May 6 in Dublin, Ireland.
Expert Perspectives
“In the setting of αβ-depleted HSCT, γδ T cells are critical effectors that provide potent graft-versus-leukemia activity without driving graft-versus-host disease,” said Dr. Alice Bertaina, MD, PhD, Co-Director of the Bass Center for Childhood Cancer and Blood Diseases at Lucile Packard Children’s Hospital, Stanford University, and lead Clinical Advisor for Laguna. “While early pharmacologic activators like zoledronic acid (ZOL) showed clinical potential, intense ZOL stimulation pushes cells into a terminally differentiated and exhausted state. Our comprehensive preclinical evaluations demonstrate that LGNA-100 drives a distinct, multifunctional γδ T cell response with improved kinetic, phenotypic, and functional features compared to ZOL, supporting more durable activation without hyperactivation or early exhaustion. I am very excited to see this translated into the clinic for these high-risk leukemia patients.”
“Pediatric AML remains one of the most challenging frontiers in oncology, demanding novel modalities that can detect and kill the disease without compounding toxicities,” said Bill Newell, former CEO of Sutro Biopharma and Strategic Advisor to Laguna. “Having spent years evaluating platforms to tackle these exact malignancies, I believe Laguna’s approach using γδ T cells to potentially solve the problems associated with high-risk leukemia is a massive leap forward. Securing this IND is a testament to the rigor of their science and positions LGNA-100 as a highly differentiated asset in the cancer immunotherapy space.”









