Quantum BioPharma Submits IND Application to FDA for Lucid-MS Phase 2 MS Trial
TORONTO, April 01, 2026 — Quantum BioPharma Ltd., a biopharmaceutical company focused on innovative assets and biotech solutions, has announced the formal submission of an Investigational New Drug application to the United States Food and Drug Administration for Lucid-21-302, known as Lucid-MS. This drug candidate is designed for the treatment of multiple sclerosis, marking a significant milestone in the company's clinical development pipeline.
Strategic Advancement in Neurological Market
This IND submission supports Quantum BioPharma's planned Phase 2 clinical trial, which will evaluate a first-in-class therapeutic treatment targeting demyelination. The move underscores the company's commitment to advancing strategic growth within the global neurological market and delivering innovative solutions for patients with debilitating neurological conditions.
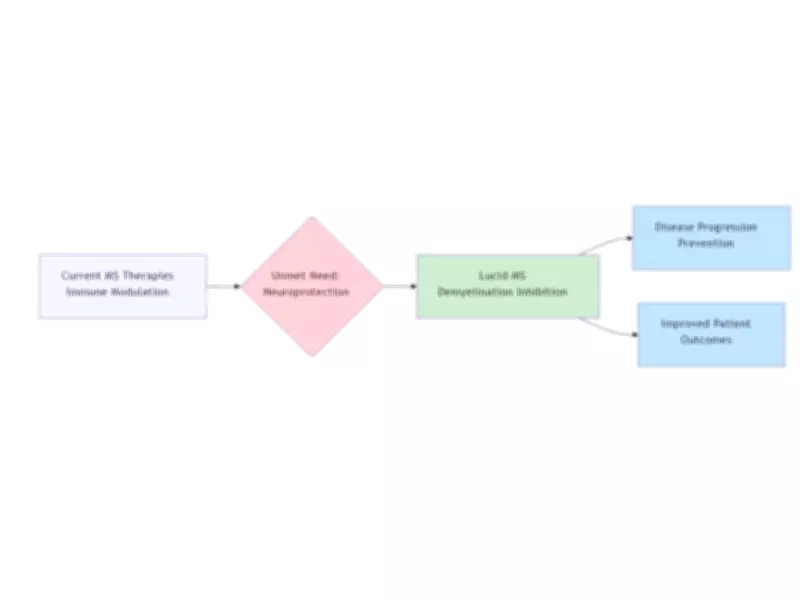
The submission represents a crucial step forward in addressing the underlying neurodegeneration in multiple sclerosis, rather than merely modulating the immune system as many existing therapies do.
Comprehensive Submission Details and Development Timeline
The IND application includes data from nonclinical pharmacology and toxicology studies, along with manufacturing and quality information, to support the initiation of the Phase 2 trial. Subject to FDA clearance, Quantum BioPharma intends to begin the study as soon as possible.
Key milestones in the development timeline include:
- Phase 1 Clinical Trials: Completed, showing the drug was safe and well-tolerated.
- IND Submission to FDA: March 2026, now submitted.
- Anticipated FDA Response: Second quarter of 2026, pending.
- Phase 2 Trial Initiation: Planned for the second quarter of 2026.
- Phase 2 Interim Data: Projected for the fourth quarter of 2026.
First-in-Class Therapeutic Innovation
Lucid-MS is engineered to provide neuroprotection by inhibiting demyelination, a key driver of disease progression in multiple sclerosis. This innovative mechanism offers a differentiated therapeutic approach in the global MS market, where current treatments often focus on immune system modulation rather than addressing neurodegeneration directly.
The planned Phase 2 trial will assess the efficacy, safety, and tolerability of Lucid-MS in individuals with MS. Previous Phase 1 trials demonstrated a favorable safety profile and good tolerability in healthy participants, laying a strong foundation for continued development.
Expert Commentary on the Submission
Dr. Andrzej Chruscinski, Vice-President of Scientific and Clinical Affairs at Quantum BioPharma, emphasized the significance of this milestone. "This IND submission is supported by a comprehensive preclinical research package designed to characterize the safety profile of Lucid-MS and support its evaluation in patients. The data we've compiled reflects many years of dedicated scientific research. We look forward to working with the FDA as we progress toward clinical evaluation of this novel approach to prevent disease progression in MS," he stated.
This development highlights Quantum BioPharma's dedication to pioneering treatments that could transform the landscape of multiple sclerosis therapy, offering hope for improved patient outcomes through targeted neuroprotective strategies.









