Biomimetic Innovations Ltd, an affiliate of Irish medical device company PBC Biomed, has announced regulatory approval for a groundbreaking clinical study of its OsStic™ injectable synthetic bone void filler. The first-in-human investigation will focus on tibial plateau fractures, complex periarticular injuries that often involve joint surface depression and loss of subchondral bone support.
Addressing Critical Surgical Challenges
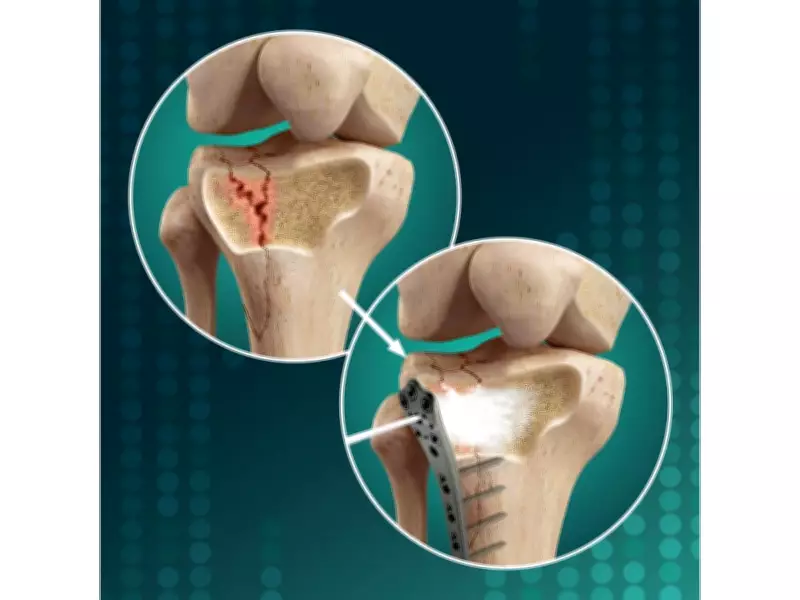
The upcoming study represents a significant advancement in orthopedic treatment options. Following elevation of the articular surface during surgery, surgeons frequently encounter metaphyseal voids that must be filled to maintain reduction and prevent secondary collapse. Existing solutions including autograft, allograft, and conventional synthetic bone substitutes have recognized limitations ranging from donor-site morbidity and inconsistent availability to brittleness and suboptimal biological integration.
Innovative Biomaterial Properties
OsStic™ has been specifically developed to overcome these challenges. The material is engineered to harden rapidly while conforming to irregular defect geometries, providing immediate mechanical support to subchondral bone. Beyond initial stabilization, the biomaterial promotes bone ingrowth and remodeling over time, addressing both short-term and long-term healing requirements.
The clinical investigation will be conducted at Leeds University Teaching Hospital in the United Kingdom under the leadership of Principal Investigator Professor Peter Giannoudis. "Along with my colleagues, I am very excited to perform this first-in-human study at Leeds Teaching Hospitals," Professor Giannoudis stated. "OsStic™ represents a 5th generation biomaterial and its unique properties are expected to revolutionize patient care and outcomes."
Comprehensive Study Objectives
The primary objective of the study focuses on evaluating device safety, including early adverse events, implant-site reactions, and device-related complications. Secondary objectives encompass multiple dimensions of patient recovery:
- Assessment of maintenance of fracture reduction
- Radiographic evidence of bone healing and remodeling
- Functional recovery measurements
- Pain outcome evaluations
- Quality of life assessments
- Exploratory gait analysis using plantar pressure mapping
Study Timeline and Significance
Patient recruitment is expected to commence in the coming weeks, with study completion anticipated by 2028. The fully funded investigation represents a crucial step in building the clinical evidence base for OsStic™ ahead of future, larger comparative trials.
Paul Burke, Managing Partner at PBC Biomed, emphasized the importance of this milestone: "We are delighted to take this next step on the OsStic™ journey with our Principal Investigator Professor Giannoudis at the Leeds University Teaching Hospital. Through our Educational Platforms and Clinical Development Board, we have now taken OsStic™ to where it can truly impact patients' lives. This is just the beginning."
Company Background
Biomimetic Innovations Ltd operates as an affiliate of PBC Biomed, a medical device company involved in design, development, manufacturing, and commercialization of innovative products. Through its advanced medical service portfolio, PBC Biomed partners with medical device, biologic, pharmaceutical, and combination product innovators to accelerate technology through product lifecycle stages.
The Shannon, Ireland-based company specializes in biomimetic materials that mimic natural biological processes, with OsStic™ representing their latest advancement in synthetic bone void filler technology. This clinical study approval by the MHRA (Medicines and Healthcare products Regulatory Agency) marks a significant regulatory milestone for the company's flagship product.